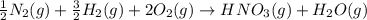Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 3h2(g) 2nh3(g) ah=-92. kj in the second step, ammonia and oxygen react to form nitric acid and water: nh3(9) + 2o2(g) → hno3(9) + h2o(g) ah=-330. kj calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest kj. пkj 1 x ś ?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:50
Working with si (metric) units for each of the following commonly used measurements, indicate its symbol. liter gram milliliter kilogram meter centigram milligram centimeter kilometer second millimeter milliseconds
Answers: 1


Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3
You know the right answer?
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen...
Questions

Mathematics, 01.07.2019 02:20


Mathematics, 01.07.2019 02:20


Mathematics, 01.07.2019 02:20

Spanish, 01.07.2019 02:20

History, 01.07.2019 02:20



History, 01.07.2019 02:20

History, 01.07.2019 02:20

Spanish, 01.07.2019 02:20

History, 01.07.2019 02:20




Biology, 01.07.2019 02:20

English, 01.07.2019 02:20


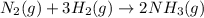 ,
, 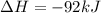
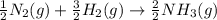 ,
, 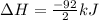
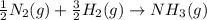 ,
, 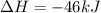 ............ (1)
............ (1)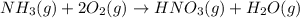
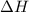 = -330 kJ ............ (2)
= -330 kJ ............ (2)