Chemistry, 14.11.2019 17:31 spreadlight10
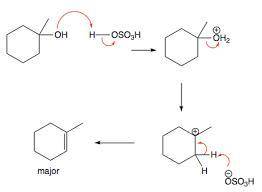
Why was it necessary to wash the crude methylcyclohexenes with aqueous sodium bicarbonate

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 20:30
Citric acid has a ph between 1 and 3. it is considered to be aa. weak acidb. weak basec. strong based. strong acid
Answers: 2
You know the right answer?
Why was it necessary to wash the crude methylcyclohexenes with aqueous sodium bicarbonate...
Questions


Business, 20.02.2020 22:51




Chemistry, 20.02.2020 22:51


History, 20.02.2020 22:51


Spanish, 20.02.2020 22:51

Computers and Technology, 20.02.2020 22:51


Mathematics, 20.02.2020 22:51

Mathematics, 20.02.2020 22:51