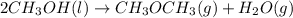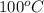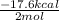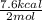Given the standard heat of combustion of methanol ch3oh is 182.6
kcal/mol, dimethyl ether ch3och3 is 347.6 kcal/mol, (methanol/
dimethyl ether in gas phase, water in liquid phase). given the heat
of vaporization of water is 10 kcal/mol, methanol is 8.4 kcal/mol,
dimethyl ether 4.8 kcal/mol. calculate the reaction of dehydration
of methanol to produce dimethyl ether. (indicate the phase of your
components).

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 16:00
How does blood clotting prevent the entry of pathogens through cuts and wounds? answer asap,, this is due tomorrow. will mark as brainliest or whatever you call it : )
Answers: 2


Chemistry, 22.06.2019 23:50
Which scientists contributed to the determination of how cfcs in clouds in the upper atmosphere could destroy ozone molecules
Answers: 1

You know the right answer?
Given the standard heat of combustion of methanol ch3oh is 182.6
kcal/mol, dimethyl ethe...
kcal/mol, dimethyl ethe...
Questions


History, 14.12.2020 23:30

Mathematics, 14.12.2020 23:30

Biology, 14.12.2020 23:30

Mathematics, 14.12.2020 23:30


English, 14.12.2020 23:30

Mathematics, 14.12.2020 23:30


History, 14.12.2020 23:30

English, 14.12.2020 23:30

Mathematics, 14.12.2020 23:30


English, 14.12.2020 23:30





History, 14.12.2020 23:30

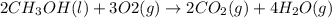 ....... (1)
....... (1)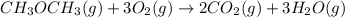 -347.6 kcal/mol
-347.6 kcal/mol
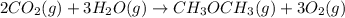 heat of reaction +347.6 kcal {sign reversed with reaction} ........ (2)
heat of reaction +347.6 kcal {sign reversed with reaction} ........ (2)