Chemistry, 13.11.2019 02:31 tjjjjjjjjjjjjjjjjjjj
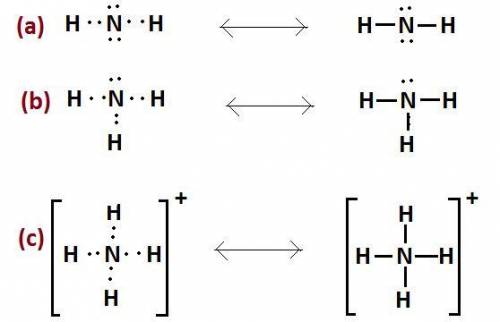
The nitrogen atom of nh2 would have the nitrogen atom of {\rm nh_2} would have blank electrons around the central nitrogen atom. electrons around the central nitrogen atom. the nitrogen atom of nh4 would have the nitrogen atom of {\rm nh_4} would have blank electrons around the central nitrogen atom. electrons around the central nitrogen atom. the nitrogen atom of nh3 would have the nitrogen atom of {\rm nh_3} would have blank electrons around the central nitrogen atom. electrons around the central nitrogen atom.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1
You know the right answer?
The nitrogen atom of nh2 would have the nitrogen atom of {\rm nh_2} would have blank electrons aroun...
Questions



English, 05.05.2020 12:11

Mathematics, 05.05.2020 12:11



Mathematics, 05.05.2020 12:11



English, 05.05.2020 12:11

History, 05.05.2020 12:11




Chemistry, 05.05.2020 12:11

Mathematics, 05.05.2020 12:11



Spanish, 05.05.2020 12:11

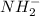 would have 8 electrons around the central nitrogen atom.
would have 8 electrons around the central nitrogen atom.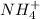 would have 8 electrons around the central nitrogen atom.
would have 8 electrons around the central nitrogen atom.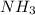 would have 8 electrons around the central nitrogen atom.
would have 8 electrons around the central nitrogen atom.