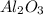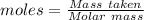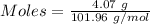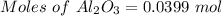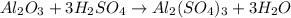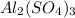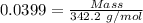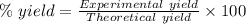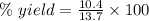Chemistry, 10.11.2019 03:31 babyboogrocks5572
For the following reaction, 4.07 grams of aluminum oxide are mixed with excess sulfuric acid. the reaction yields 10.4 grams of aluminum sulfate. aluminum oxide (s) + sulfuric acid (aq) aluminum sulfate (aq) + water (l) what is the theoretical yield of aluminum sulfate? grams what is the percent yield for this reaction? %

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Strong conductivity of plasma allows it to act and react as and
Answers: 2

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 22:30
Which of the following is true about the speed of light? it depends on the wavelength.
Answers: 3
You know the right answer?
For the following reaction, 4.07 grams of aluminum oxide are mixed with excess sulfuric acid. the re...
Questions

History, 18.02.2022 01:10

Mathematics, 18.02.2022 01:10

Computers and Technology, 18.02.2022 01:10





Mathematics, 18.02.2022 01:20

English, 18.02.2022 01:20

Mathematics, 18.02.2022 01:20



Chemistry, 18.02.2022 01:20




Mathematics, 18.02.2022 01:20

Computers and Technology, 18.02.2022 01:20