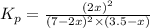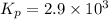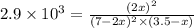Chemistry, 06.11.2019 04:31 PlsHelpMeh3401
Calculate the pressures of no, cl2, and nocl in an equilibrium mixture produced by the reaction of a starting mixture with 7.0 atm no and 3.5 atm cl2. (hint: kp is relatively large; assume the reaction goes to completion then comes back to equilibrium.)
2 no(g) + cl2(g) --> 2 nocl(g)kp = 2.9 ✕ 103 at 149°c

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural.question 2 reflects a moral or social value.question 3 refers to something that can be measured.question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 22.06.2019 15:10
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1

Chemistry, 22.06.2019 16:00
Answer asap : ( a. how does mucus prevent the entry of pathogens? b. describe two ways white blood cells protect us from pathogens.
Answers: 1
You know the right answer?
Calculate the pressures of no, cl2, and nocl in an equilibrium mixture produced by the reaction of a...
Questions


English, 07.09.2019 01:10


English, 07.09.2019 01:10

English, 07.09.2019 01:10

History, 07.09.2019 01:10

Health, 07.09.2019 01:10

Mathematics, 07.09.2019 01:10

Mathematics, 07.09.2019 01:10

Physics, 07.09.2019 01:10








Computers and Technology, 07.09.2019 01:10


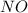 ,
,  , and
, and 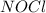 in an equilibrium mixture are 0.4 atm , 0.2 atm and 6.6 atm respectively.
in an equilibrium mixture are 0.4 atm , 0.2 atm and 6.6 atm respectively.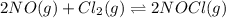
![K_p=\frac{[NOCl]^2}{[NO]^2[Cl_2]}](/tpl/images/0361/4899/09f8c.png)