Chemistry, 05.11.2019 06:31 issagirl05
A23.0-ml volume of o2, collected over water at 752 torr and 22â°c, is produced from the thermal decomposition of kclo3. the vapor pressure of water at 22â°c is 19.8 torr. how many moles of o2 are collected?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 05:20
Temperature is _related to the average kinetic energy of a gas. inversely directly not disproportionally
Answers: 1

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 17:30
Energy defines the different "states" of matter. in no more than 3 sentences, describe the amount of kinetic energy that each of the 3 states of matter possesses and relate that to the atom/molecular motion of each "state".
Answers: 2
You know the right answer?
A23.0-ml volume of o2, collected over water at 752 torr and 22â°c, is produced from the thermal deco...
Questions



Mathematics, 20.11.2021 05:20



Computers and Technology, 20.11.2021 05:20







Mathematics, 20.11.2021 05:30

Geography, 20.11.2021 05:30






Mathematics, 20.11.2021 05:30

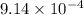 moles
moles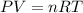
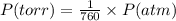
![22^oC=[22+273]K=295K](/tpl/images/0360/1433/7919f.png)