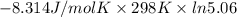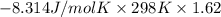Chemistry, 05.11.2019 00:31 jessemartinez1
For the reaction a(g) + 2 b(g) ↔ c(g) the initial partial pressures of gases a, b, and c are all 0.109 atm. once equilibrium has been established, it is found that pc = 0.047 atm. what is δg° for this reaction (in kj/mol) at 25°c?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:40
Which of the following pressures is equal to 760 mm hg? 2.0 atm 101.3 pa 101,300 kpa 101,300 pa
Answers: 2


Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 13:00
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3
You know the right answer?
For the reaction a(g) + 2 b(g) ↔ c(g) the initial partial pressures of gases a, b, and c are all 0.1...
Questions

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Spanish, 17.09.2020 14:01

English, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Biology, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

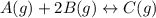
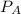 = 0.109 atm,
= 0.109 atm, 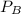 = 0.109 atm,
= 0.109 atm,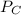 = 0.109 atm
= 0.109 atm![[0.109 + (2 \times 0.062)]](/tpl/images/0359/5284/f1aca.png) atm
atm 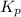 as follows.
as follows.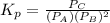
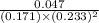
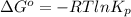
 as follows.
as follows.