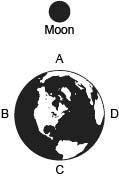
Chemistry, 03.11.2019 22:31 jasminetaylor4587
Using 25.0cm3 of aqueous lithium hydroxide, concentration 2.48mol/dm3, 2.20g of hydrated lithium sulfate was obtained. calculate the percentage yield, giving your answer to one decimal place.
2lioh h2so4 → li2so4 2h2o li2so4 h2o → li2so4.h2o
the number of moles of lioh used =
the number of moles of li2so4.h2o which could be formed =
mass of one mole of li2so4.h2o = 128 g
the maximum yield of li2so4.h2o =
percentage yield = % [4]

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:10
The peak wavelength for the blackbody curve of a star is in the uv range. assuming the radiation from this star can reach earth, would you be able to see it?
Answers: 2

Chemistry, 22.06.2019 13:30
Mary is conducting an experiment on how pollution affects plant growth. how can she ensure that her data are reliable?
Answers: 3

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1

Chemistry, 22.06.2019 17:30
What type of organic molecule comprises the majority of a potato?
Answers: 1
You know the right answer?
Using 25.0cm3 of aqueous lithium hydroxide, concentration 2.48mol/dm3, 2.20g of hydrated lithium sul...
Questions


Social Studies, 21.08.2019 19:50

Health, 21.08.2019 19:50


Physics, 21.08.2019 19:50








Mathematics, 21.08.2019 19:50





Biology, 21.08.2019 19:50

History, 21.08.2019 19:50