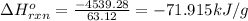Pentaborane−9 (b5h9) is a colorless, highly reactive liquid that will burst into flames when exposed to oxygen. the reaction is 2b5h9(l) + 12o2(g) → 5b2o3(s) + 9h2o(l) calculate the kilojoules of heat released per gram of the compound reacted with oxygen. the standard enthalpy of formations of b5h9(l), b2o3(s), and h2o(l) are 73.2, −1271.94, and −285.83 kj/mol, respectively.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
What’s the scientific notation for the number 6,840,000,000
Answers: 1

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1
You know the right answer?
Pentaborane−9 (b5h9) is a colorless, highly reactive liquid that will burst into flames when exposed...
Questions





Mathematics, 13.08.2021 04:20



Computers and Technology, 13.08.2021 04:20





Computers and Technology, 13.08.2021 04:20

Mathematics, 13.08.2021 04:20




Social Studies, 13.08.2021 04:20

Mathematics, 13.08.2021 04:20

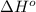
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_f(product)]-\sum [n\times \Delta H^o_f(reactant)]](/tpl/images/0352/3055/45485.png)
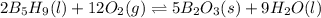
![\Delta H^o_{rxn}=[(n_{(B_2O_3)}\times \Delta H^o_f_{(B_2O_3)})+(n_{(H_2O)}\times \Delta H^o_f_{(H_2O)})]-[(n_{(B_5H_9)}\times \Delta H^o_f_{(B_5H_9)})+(n_{(O_2)}\times \Delta H^o_f_{(O_2)})]](/tpl/images/0352/3055/0889c.png)
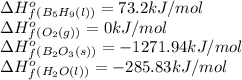
![\Delta H^o_{rxn}=[(5\times -1271.94)+(9\times -285.83)]-[(2\times 73.2)+(12\times 0)]=-9078.57kJ/mol](/tpl/images/0352/3055/ac167.png)
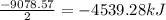
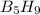 = 63.12 g/mole
= 63.12 g/mole