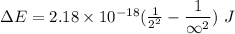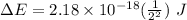Chemistry, 30.10.2019 05:31 htiffany0225
Calculate the energy, in joules, required to ionize a hydrogen atom when its electron is initially in the n =2 energy level. the energy needed to ionize a ground-state hydrogen atom is 2.18 x 10–18 j.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 22.06.2019 18:30
The famous scientist galileo galilei did several experiments with sloping planes, which he rolled metal balls down so that he could study motion. by changing the slope, he could study how the speed at which the ball rolled was affected. what was the independent variable in galileo's experiment? a. the speed of the ball b. the slope of the plane c. whether the ball moved d. what the ball was made of
Answers: 2

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
Calculate the energy, in joules, required to ionize a hydrogen atom when its electron is initially i...
Questions


Mathematics, 18.03.2021 01:00

Physics, 18.03.2021 01:00


English, 18.03.2021 01:00

History, 18.03.2021 01:00



Biology, 18.03.2021 01:00




Mathematics, 18.03.2021 01:00

English, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

History, 18.03.2021 01:00


Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

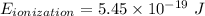
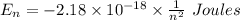
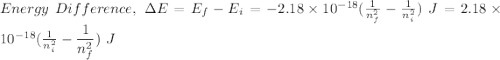
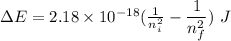
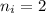 and
and 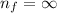 (As the hydrogen has to ionize)
(As the hydrogen has to ionize)