
Chemistry, 24.10.2019 17:43 odetteerivera
In the process for the manufacture of chlorine by direct oxidation of hcl with air over a catalyst to form cl2 and h2o (only), the exit product is composed of mole fractions of cl2 (21.4%), h2o (21.4%), and n2 (45.0%) – the remainder is hcl and o2. calculate and /or identify: (a) the limiting reactant? (b) the percentage excess reactant? (c) the degree of completion of the reaction? (d) the extent of reaction?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 18:00
Many pharmaceutical drugs are organic compounds that were originally synthesized in the laboratory. which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 2

Chemistry, 22.06.2019 22:00
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1

You know the right answer?
In the process for the manufacture of chlorine by direct oxidation of hcl with air over a catalyst t...
Questions


English, 31.08.2019 16:00




History, 31.08.2019 16:00


Mathematics, 31.08.2019 16:00


History, 31.08.2019 16:00

Biology, 31.08.2019 16:00

Health, 31.08.2019 16:00

Computers and Technology, 31.08.2019 16:00

English, 31.08.2019 16:00


History, 31.08.2019 16:00

Mathematics, 31.08.2019 16:00

English, 31.08.2019 16:00


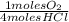 = 0,10725 moles of O₂
= 0,10725 moles of O₂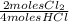 = 0,2145 moles of Cl₂ that are the same than moles of H₂O.
= 0,2145 moles of Cl₂ that are the same than moles of H₂O.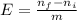
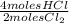 = 0,001 moles of HCl. As initial moles are 0,429 and stoichiometric coefficient is 4, extent of reaction is 0,107
= 0,001 moles of HCl. As initial moles are 0,429 and stoichiometric coefficient is 4, extent of reaction is 0,107

