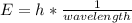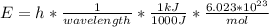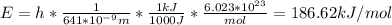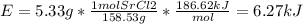Chemistry, 23.10.2019 17:00 camirialchambers17
In fireworks, the heat of the reaction of an oxidizing agent, such as kclo4, with an organic compound excites certain salts, which emit specific colors. strontium salts have an intense emission at 641 nm. what is the energy (in kj) of this emission for 5.33 g of the chloride salt of strontium? assume that all the heat produced is converted to emitted light. enter to 2 decimal places.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 17:30
What type of organic molecule comprises the majority of a potato?
Answers: 1

Chemistry, 22.06.2019 19:20
15. which of the following is not human-caused groundwater pollution? a. water in an aquifer dissolves elements such as arsenic and mercury from surrounding rock. b. water in an aquifer is contaminated by leachate that seeps into the ground from a landfill. c. water in an aquifer becomes polluted with chemicals used in hydraulic fracturing, or fracking. d. water in an aquifer absorbs harmful bacteria from the drainage field of a septic tank.
Answers: 1
You know the right answer?
In fireworks, the heat of the reaction of an oxidizing agent, such as kclo4, with an organic compoun...
Questions

English, 22.01.2021 23:30

Mathematics, 22.01.2021 23:30

Mathematics, 22.01.2021 23:30



History, 22.01.2021 23:30

History, 22.01.2021 23:30


Mathematics, 22.01.2021 23:30


Mathematics, 22.01.2021 23:30




Mathematics, 22.01.2021 23:30


Social Studies, 22.01.2021 23:30

Mathematics, 22.01.2021 23:30

Chemistry, 22.01.2021 23:30

Chemistry, 22.01.2021 23:30