
Chemistry, 16.10.2019 21:30 itzdulceee
Ph is a logarithmic scale used to indicate the hydrogen ion concentration, [h+], of a solution: ph=−log[h+] due to the autoionization of water, in any aqueous solution, the hydrogen ion concentration and the hydroxide ion concentration, [oh−], are related to each other by the kw of water: kw=[h+][oh−]=1.00×10−14 where 1.00×10−14 is the value at approximately 297 k. based on this relation, the ph and poh are also related to each other as 14.00=ph+poh the temperature for each solution is carried out at approximately 297 k where kw=1.00×10−14. part a 0.35 g of hydrogen chloride (hcl) is dissolved in water to make 2.5 l of solution. what is the ph of the resulting hydrochloric acid solution?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
What is the result of multiplying (2.5 × 1010) × (2.0 × 10-7)? a. 5.0 × 103 b. 5.0 × 10-3 c. 5.0 × 1017 d. 5.0 × 10-17
Answers: 1


Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
You know the right answer?
Ph is a logarithmic scale used to indicate the hydrogen ion concentration, [h+], of a solution: ph=...
Questions

Mathematics, 11.09.2020 03:01

Computers and Technology, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Computers and Technology, 11.09.2020 03:01

Computers and Technology, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Biology, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

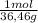 = 9,6x10⁻³ moles HCl ≡ moles H⁺
= 9,6x10⁻³ moles HCl ≡ moles H⁺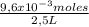 = 3,84x10⁻³M
= 3,84x10⁻³M

