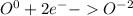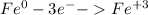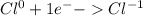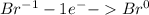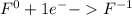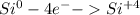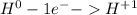Chemistry, 16.10.2019 17:30 postorivofarms
For the complete redox reactions given here, write the half-reactions and identify the oxidizing and re- ducing agents: (a) 4fe 3o2 ¡ 2fe2o3
(b) cl2 2nabr ¡ 2nacl br2 (c)si2f2¡sif4 (d) h2 cl2 ¡ 2hc

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Which compounds have the empirical formula ch2o? a.c2h4o2 b.c3h6o3 c.ch2o2 d.c5h10o5 e.c6h12o6
Answers: 3

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

Chemistry, 22.06.2019 22:00
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2

Chemistry, 22.06.2019 23:30
To find the work done, the force exerted and distance moved are multiplied. a couch is moved twice before you are happy with its placement. the same force was used to move the couch both times. if more work is done the first time it is moved, what do you know about the distance it was moved? a) when more work was done, the couch was moved the same distance. b) when more work was done, the couch was moved less. c) when more work was done, the couch was moved further. d) when more work was done, the couch wasn't moved at all.
Answers: 1
You know the right answer?
For the complete redox reactions given here, write the half-reactions and identify the oxidizing and...
Questions


Mathematics, 01.12.2021 05:30

Mathematics, 01.12.2021 05:30


Engineering, 01.12.2021 05:30



Mathematics, 01.12.2021 05:30

Computers and Technology, 01.12.2021 05:30


Mathematics, 01.12.2021 05:30

Mathematics, 01.12.2021 05:30

English, 01.12.2021 05:30







Mathematics, 01.12.2021 05:30