
Chemistry, 11.10.2019 22:30 adhanom1271
Magnesium (used in the manufacture of light alloys) reacts with iron(iii) chloride to form magnesium chloride and iron. 3mg(s) + 2fecl₃(s) → 3mgcl₂(s) + 2fe(s) a mixture of 41.0 g of magnesium ( = 24.31 g/mol) and 175 g of iron(iii) chloride ( = 162.2 g/mol) is allowed to react. identify the limiting reactant and determine the mass of the excess reactant present in the vessel when the reaction is complete.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
50 pts plz what is the physical state of matter of baking soda.
Answers: 1

Chemistry, 22.06.2019 20:00
Which of the following would not diffuse through the plasma membrane by means of simple diffusion? 1 oxygen 2 glucose 3 a steroid hormone 4 a lipid soluble vitamin
Answers: 3

Chemistry, 23.06.2019 03:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1

You know the right answer?
Magnesium (used in the manufacture of light alloys) reacts with iron(iii) chloride to form magnesium...
Questions



Biology, 25.07.2019 05:30

History, 25.07.2019 05:30


Health, 25.07.2019 05:30

Physics, 25.07.2019 05:30


Mathematics, 25.07.2019 05:30

Mathematics, 25.07.2019 05:30



Health, 25.07.2019 05:30


Mathematics, 25.07.2019 05:30

Mathematics, 25.07.2019 05:30

Chemistry, 25.07.2019 05:30



Mathematics, 25.07.2019 05:30

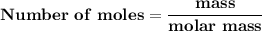
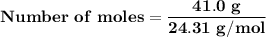
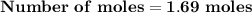
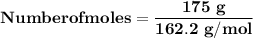
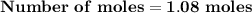
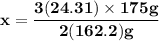 x = 39.34 grams
x = 39.34 grams

