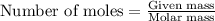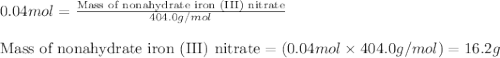In reality, a hydrate of iron(iii) nitrate had to be used, not the anhydrous salt. as you may guess, some of the hydrate’s mass is water, and some is iron(iii) nitrate. how many grams of fe(no3)3•9h2o needed to be dissolved in water to make 2 l of 0.0020 m fe(no3)3? molecular weight of the nonahydrate is 404.0 g/mol. hint: try to set up an equation using x, and solving it. assume that the density of your solution is 1.000 g/ml.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1


Chemistry, 22.06.2019 23:10
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 3
You know the right answer?
In reality, a hydrate of iron(iii) nitrate had to be used, not the anhydrous salt. as you may guess,...
Questions

English, 28.01.2021 03:50

History, 28.01.2021 03:50

English, 28.01.2021 03:50





Mathematics, 28.01.2021 03:50

Mathematics, 28.01.2021 03:50

Biology, 28.01.2021 03:50

English, 28.01.2021 03:50









Biology, 28.01.2021 03:50

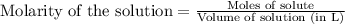
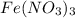 = 0.0020 M
= 0.0020 M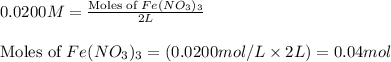
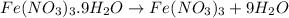
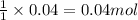 of hydrated iron (III) nitrate
of hydrated iron (III) nitrate