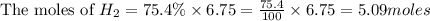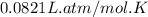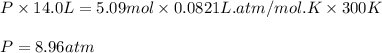Chemistry, 07.10.2019 23:00 brandytyler317fries
Consider 2 al + 6 hcl → 2 alcl3 + 3 h2 , the reaction of al with hcl to produce hydrogen gas. what is the pressure of h2 if the hydrogen gas collected occupies 14.0 l at 300.k and was produced upon reaction of 4.50 moles of al and excess hcl in a process that has a 75.4 percent yield?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 04:40
Listen base your answer to the question on the information below.propane is a fuel that is sold in rigid, pressurized cylinders. most of the propane in a cylinder is liquid, with gas in the space above the liquid level. when propane is released from the cylinder, the propane leaves the cylinder as a gas. propane gas is used as a fuel by mixing it with oxygen in the air and igniting the mixture, as represented by the balanced equation below.c3h8(g) + 5o2(g) → 3co2(g) + 4h2o() + 2219.2 kja small amount of methanethiol, which has a distinct odor, is added to the propane to consumers detect a propane leak. in methanethiol, the odor is caused by the thiol functional group (–sh). methanethiol, ch3sh, has a structure that is very similar to the structure of methanol.what is the correct structural formula for a molecule of methanethiol
Answers: 3

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1
You know the right answer?
Consider 2 al + 6 hcl → 2 alcl3 + 3 h2 , the reaction of al with hcl to produce hydrogen gas. what i...
Questions







Biology, 05.11.2020 06:10


Social Studies, 05.11.2020 06:10


Mathematics, 05.11.2020 06:10






Social Studies, 05.11.2020 06:10



History, 05.11.2020 06:10

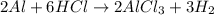
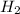 gas
gas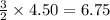 moles of
moles of