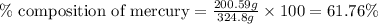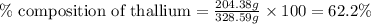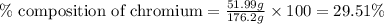Chemistry, 07.10.2019 20:00 hala201490
Be sure to answer all parts. dimercaprol (hsch2chshch2oh) was developed during world war i as an antidote to arsenic-based poison gas and is used today to treat heavy-metal poisoning. it binds the toxic element and carries it out of the body. (a) if each molecule binds one arsenic (as) atom, how many atoms of as could be removed by 696 mg of dimercaprol? × 10 atoms as (enter your answer in scientific notation.) (b) if one molecule binds one metal atom, calculate the mass % of each of the following metals in a metal-dimercaprol combination: mercury, thallium, chromium.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Acar tire has a pressure of 2.38 atm at 15.2 c. if the pressure inside reached 4.08 atm, the tire will explode. how hot would the tire have to get for this to happen? report the temperature in degrees celsius.
Answers: 2

Chemistry, 21.06.2019 19:10
Nuclear fusion is the source of energy for stars. besides hydrogen, which other element is most likely also common in stars?
Answers: 1


Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2
You know the right answer?
Be sure to answer all parts. dimercaprol (hsch2chshch2oh) was developed during world war i as an ant...
Questions

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Biology, 14.09.2020 04:01

History, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

English, 14.09.2020 04:01

Biology, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

English, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Physics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

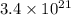
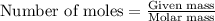
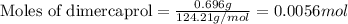
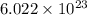 number of molecules.
number of molecules.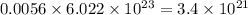 number of molecules.
number of molecules.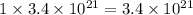 number of arsenic atoms
number of arsenic atoms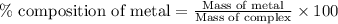 ......(1)
......(1)