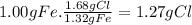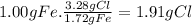Prelab for experiment 4: the atomic theory of matter 1. demonstrate the law of multiple proportions with the following data: a. iron forms two compounds with chlorine, compound x and compound y. when 3.00 g of compound x was analyzed, it was found that 1.68 g of ci combined with 1.32 g of fe. when 5.00 g of compound y was analyzed, it was found that 3.28 g of cl combined with 1.72 g of fe. to illustrate the law, one needs to know how much of one element combines with the same mass as the other element in each compound, but the data above give different masses for both fe and cl. one way to standardize the data is to find out how much of one element, say cl, combines with (if 1.75 g of cl combines with 1.25 g of fe, how many g of cl would combine with 1.00 g of ") do this for both compounds, x and y exactly 1 g of the other, say fe. this can be done by a simple proportion method

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 09:30
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

You know the right answer?
Prelab for experiment 4: the atomic theory of matter 1. demonstrate the law of multiple proportions...
Questions

English, 15.07.2021 20:40


Mathematics, 15.07.2021 20:40

Mathematics, 15.07.2021 20:40

Mathematics, 15.07.2021 20:40



Mathematics, 15.07.2021 20:40



Mathematics, 15.07.2021 20:40

History, 15.07.2021 20:40

Mathematics, 15.07.2021 20:40


Social Studies, 15.07.2021 20:50

Mathematics, 15.07.2021 20:50

Mathematics, 15.07.2021 20:50