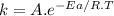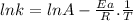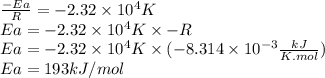Chemistry, 05.10.2019 04:20 Dreynolds1667
For the gas phase isomerization of cis-cyanostyrene, cis-c6h5ch=chc --> ntrans-c6h5ch=chcn the rate constant has been determined at several temperatures. when ln k in s-1 is plotted against the reciprocal of the kelvin temperature, the resulting linear plot has a slope of -2.32×104 k and a y-intercept of 26.7. the activation energy for the gas phase isomerization of cis-cyanostyrene is kj/mol.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 11:00
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow.part ao2-(aq)+h2o(l)< => express your answer as part of a chemical equation. identify all of the phases in your answer.o2-(aq)+h2o(l) < => oh-(aq)+oh-(aq)part bpredict whether the equilibrium lies to the left or to the right of the equation in previous part.h2o is a stronger acid than oh–, so the equilibrium lies to the right.h2o is a weaker acid than oh–, so the equilibrium lies to the left.h2o is a stronger acid than oh–, so the equilibrium lies to the left.h2o is a weaker acid than oh–, so the equilibrium lies to the right.part cch3cooh(aq)+hs? (aq) < => express your answer as part of a chemical equation. identify all of the phases in your answer.ch3cooh(aq)+hs-(aq) < => h2s(aq)+c2h3o2-(aq)h2s(aq)+c2h3o2-(aq)part dpredict whether the equilibrium lies to the left or to the right of the equation in previous part.ch3cooh is a weaker acid than h2s, so the equilibrium lies to the right.ch3cooh is a weaker acid than h2s, so the equilibrium lies to the left.ch3cooh is a stronger acid than h2s, so the equilibrium lies to the right.ch3cooh is a stronger acid than h2s, so the equilibrium lies to the left.part eno2-(aq)+h2o(l) < => express your answer as part of a chemical equation. identify all of the phases in your answer.no2-(aq)+h2o(l) < => part fpredict whether the equilibrium lies to the left or to the right of the equation in previous part.hno2 is a stronger acid than h2o, so the equilibrium lies to the right.hno2 is a weaker acid than h2o, so the equilibrium lies to the left.hno2 is a stronger acid than h2o, so the equilibrium lies to the left.hno2 is a weaker acid than h2o, so the equilibrium lies to the right.
Answers: 1

Chemistry, 23.06.2019 13:00
Aecosystem is if it can continue to function over long periods of time
Answers: 1

Chemistry, 23.06.2019 17:20
Which determines the state of matter for any material? a. the size of the atoms b. the hardness of the material c. the behavior of the molecules d. the weight of the material b. fixed volume, takes the shape of its container c. completely fills its container, definite shape d. completely fills its container, takes the shape of its container
Answers: 1
You know the right answer?
For the gas phase isomerization of cis-cyanostyrene, cis-c6h5ch=chc --> ntrans-c6h5ch=chcn the r...
Questions

Mathematics, 10.11.2020 05:20

Biology, 10.11.2020 05:20

History, 10.11.2020 05:20

Mathematics, 10.11.2020 05:20



Biology, 10.11.2020 05:20





Law, 10.11.2020 05:30

Computers and Technology, 10.11.2020 05:30

Social Studies, 10.11.2020 05:30



Biology, 10.11.2020 05:30

Engineering, 10.11.2020 05:30


Mathematics, 10.11.2020 05:30