
Chemistry, 06.10.2019 04:00 zwbaby3693
At 298 k, the equilibrium concentration of o2 is 1.6 x 10-2 m, and the equilibrium concentration of o3 is 2.86 x 10-28 m. what is the equilibrium constant of the reaction at this temperature?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:10
Nitrogen (n), phosphorus (p), and potassium (k) are the main nutrients in plant fertilizers. according to an industry convention, the numbers on the label refer to the mass percents of n, p2o5, and k2o, in that order. calculate the n: p: k ratio of a 30: 10: 10 fertilizer in terms of moles of each element, and express it as x: y: 1.0.
Answers: 1

Chemistry, 22.06.2019 08:00
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3
You know the right answer?
At 298 k, the equilibrium concentration of o2 is 1.6 x 10-2 m, and the equilibrium concentration of...
Questions

Social Studies, 07.09.2020 14:01



Mathematics, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01

English, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01

Computers and Technology, 07.09.2020 14:01

Arts, 07.09.2020 14:01


Mathematics, 07.09.2020 14:01

History, 07.09.2020 14:01

Biology, 07.09.2020 14:01


English, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01

Spanish, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01

Health, 07.09.2020 14:01


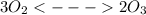
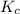 is the equilibrium constant and is defined as products concentration over reactant concentration and the coefficient is raised to its power. Thus we have the
is the equilibrium constant and is defined as products concentration over reactant concentration and the coefficient is raised to its power. Thus we have the ![K_c = \frac {[Products concentration]}{[Reactants concentration]}](/tpl/images/0291/9821/c00b5.png)
![K_c =\frac {[O_3 ]^2}{[O_2 ]^3}](/tpl/images/0291/9821/20678.png)
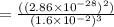
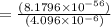
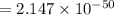 (Answer)
(Answer)

