Consider the following reversible reaction.
c(s)+o2(g)< —> co2(g)
what is the equi...

Chemistry, 02.10.2019 23:10 makayladishon
Consider the following reversible reaction.
c(s)+o2(g)< —> co2(g)
what is the equilibrium constant expression for the given system?
[co]
[co]
[co]

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Which is the layer underground where all empty spaces are filled with a combination of air and water ?
Answers: 1

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1


Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1
You know the right answer?
Questions


Medicine, 22.01.2021 19:40

Chemistry, 22.01.2021 19:40


Business, 22.01.2021 19:40


Business, 22.01.2021 19:40





Business, 22.01.2021 19:40

Mathematics, 22.01.2021 19:40

Mathematics, 22.01.2021 19:40


Mathematics, 22.01.2021 19:40

Chemistry, 22.01.2021 19:40



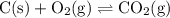
![K_{eq} = \dfrac{[\text{Products}]}{[\text{Reactants}]}](/tpl/images/0284/4834/57030.png)
![K_{eq} = \dfrac{[\text{CO}_{2}]} {[\text{O}_{2}]}](/tpl/images/0284/4834/a61e4.png)



