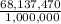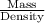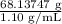Chemistry, 02.10.2019 20:00 ewalchloe5067920
Asolution used to chlorinate a home swimming pool contains 7% chlorine by mass. an ideal chlorine level for the pool is one part per million (1 ppm). (think of 1 ppm as being 1 g chlorine per million grams of water). if you assume densities of 1.10 g/ml for the chlorine solution and 1.00 g/ml for the swimming pool water, what volume of the chlorine solution, in liters, is required to produce a chlorine level of 1 ppm in an 18,000 gal swimming pool?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5 m hcl? show all of the work needed to solve this problem. mg (s) + 2hcl (aq) → mgcl2 (aq) + h2 (g)
Answers: 3

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2
You know the right answer?
Asolution used to chlorinate a home swimming pool contains 7% chlorine by mass. an ideal chlorine le...
Questions

Mathematics, 19.08.2019 14:30



Social Studies, 19.08.2019 14:30




Mathematics, 19.08.2019 14:30

Mathematics, 19.08.2019 14:30



Mathematics, 19.08.2019 14:30


Biology, 19.08.2019 14:30

History, 19.08.2019 14:30





Mathematics, 19.08.2019 14:30