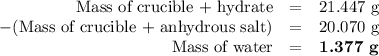Analo387: percent water in a hydrate
3. the mass of a crucible and a hydrated salt was found...

Chemistry, 02.10.2019 17:30 abdullaketbi71
Analo387: percent water in a hydrate
3. the mass of a crucible and a hydrated salt was found to be 21.447 g. the mass of the crucible and the
anhydrous salt was 20.070 g. the mass of the crucible was 17.985 g.
(1) calculate the mass of the hydrate heated.
(2) calculate the mass of water lost from the hydrate during heating.
(3) calculate the percent water in the hydrate.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Which statement describes evidence of a chemical reaction? a) ice melting eliminate b) water boiling c) lighting a match d) grape juice freezing
Answers: 3

Chemistry, 21.06.2019 22:30
Hot air balloons float in the air because of the difference in density between cold and hot air. in this problem, you will estimate the minimum temperature the gas inside the balloon needs to be, for it to take off. to do this, use the following variables and make these assumptions: the combined weight of the pilot basket together with that of the balloon fabric and other equipment is w. the volume of the hot air inside the balloon when it is inflated is v. the absolute temperature of the hot air at the bottom of the balloon is th (where th> tc). the absolute temperature of the cold air outside the balloon is tc and its density is ďc. the balloon is open at the bottom, so that the pressure inside and outside the balloon is the same. as always, treat air as an ideal gas. use g for the magnitude of the acceleration due to gravity.
Answers: 1


Chemistry, 23.06.2019 00:00
#20 which type of bond is formed when bases pair in dna? ionic bond covalent bond coordinate bond hydrogen bond
Answers: 1
You know the right answer?
Questions


English, 05.11.2020 18:10

Mathematics, 05.11.2020 18:10

Mathematics, 05.11.2020 18:10

Mathematics, 05.11.2020 18:10




Biology, 05.11.2020 18:10

Arts, 05.11.2020 18:10

Mathematics, 05.11.2020 18:10

Computers and Technology, 05.11.2020 18:10

Social Studies, 05.11.2020 18:10


Mathematics, 05.11.2020 18:10


French, 05.11.2020 18:10

Chemistry, 05.11.2020 18:10