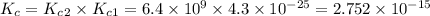Chemistry, 01.10.2019 23:00 morgaaaan651
The following reactions have the indicated equilibrium constants at a particular temperature: n2(g) + o2(g) ⇌ 2no(g)kc = 4.3 × 10−25 2no(g) + o2(g) ⇌ 2no2(g)kc = 6.4 × 109 determine the value of the equilibrium constant for the following equation at the same temperature: n2(g) + 2o2(g) ⇌ 2no2(g) kc = × 10 (enter your answer in scientific notation.)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1

Chemistry, 22.06.2019 20:00
What happens to the temperature of a substance when the average kinetic energy of its particles increases?
Answers: 3

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
The following reactions have the indicated equilibrium constants at a particular temperature: n2(g)...
Questions

Mathematics, 01.05.2021 01:00


Mathematics, 01.05.2021 01:00




Mathematics, 01.05.2021 01:00

Mathematics, 01.05.2021 01:00



Chemistry, 01.05.2021 01:00


Mathematics, 01.05.2021 01:00

Mathematics, 01.05.2021 01:00




Business, 01.05.2021 01:00

Mathematics, 01.05.2021 01:00

Mathematics, 01.05.2021 01:00

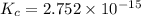
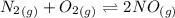
![K_c_1=\frac {[NO]^2}{[N_2][O_2]}=4.3\times 10^{-25}](/tpl/images/0281/1482/cff32.png)
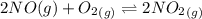
![K_c_2=\frac {[NO_2]^2}{[NO]^2[O_2]}=6.4\times 10^{9}](/tpl/images/0281/1482/9df1f.png)
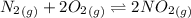
![K_c=\frac {[NO_2]^2}{[N_2][O_2]^2}](/tpl/images/0281/1482/1467b.png)
![[NO]^2](/tpl/images/0281/1482/b4f90.png) and rearranging in the above equation as:
and rearranging in the above equation as:![K_c=\frac {[NO_2]^2}{[NO]^2[O_2]}\times \frac {[NO]^2}{[N_2][O_2]}](/tpl/images/0281/1482/822cf.png)