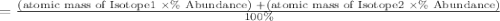Chemistry, 01.10.2019 06:30 trentvw1174
Afictitious element q has two naturally occurring isotopes. the first isotope has an abundance of 20.00% and a mass of 39.96 amu and the second isotope of element q has an abundance of 80.00% and a mass of 43.39amu. calculate the weighted atomic mass of element q to the nearest tenth. to earn credit, be sure to show the work that leads to your answer.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 19:00
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3
You know the right answer?
Afictitious element q has two naturally occurring isotopes. the first isotope has an abundance of 20...
Questions


English, 12.04.2020 17:10

English, 12.04.2020 17:10














History, 12.04.2020 17:26

English, 12.04.2020 17:26

Mathematics, 12.04.2020 17:26