
Chemistry, 01.10.2019 04:10 bettybales1986
A26.08 g mixture of zinc and sodium is reacted with a stoichiometric amount of sulfuric acid. the reaction mixture is then reacted with 132 ml of 3.80 m barium chloride to produce the maximum possible amount of barium sulfate. determine the percent sodium by mass in the original mixture.

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 04:31
How many grams of iron can be made from 16.5 grams of fe2o3
Answers: 1

Chemistry, 23.06.2019 12:30
You have 125 g of a certain seasoning and are told that it contains 70.0 g of salt. what is the peroentage of salt by mass in this seasoning?
Answers: 2

Chemistry, 23.06.2019 13:00
If volume remains the same while the mass of a substance the density of the substance
Answers: 1

Chemistry, 23.06.2019 22:30
Write the formula for the conjugate base acid of hexanoic acid
Answers: 1
You know the right answer?
A26.08 g mixture of zinc and sodium is reacted with a stoichiometric amount of sulfuric acid. the re...
Questions



Mathematics, 19.04.2021 19:10


Social Studies, 19.04.2021 19:10


Mathematics, 19.04.2021 19:10

History, 19.04.2021 19:10


English, 19.04.2021 19:10


Mathematics, 19.04.2021 19:10

English, 19.04.2021 19:10



Mathematics, 19.04.2021 19:10

Engineering, 19.04.2021 19:10

Mathematics, 19.04.2021 19:10

Chemistry, 19.04.2021 19:10

History, 19.04.2021 19:10

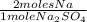 × 22,99 g/mole = 23,08 g of Na
× 22,99 g/mole = 23,08 g of Na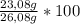 = 88,50%
= 88,50% 

