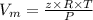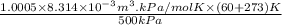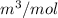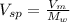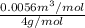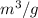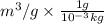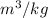Chemistry, 25.09.2019 03:00 scottcounts757
Calculate the specific volume of helium using the compressibility factor. 500 kpa, 60 degrees celsius

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:10
Describe the difference between a. a hypothesis and a theory and b. an observation and an experiment.
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 23.06.2019 05:40
Why is it incorrect to balance a chemical equation by changing the subscripts? explain.
Answers: 2

Chemistry, 23.06.2019 08:40
Which statement is true according to the kinetic theory? a. molecules of different gases with the same mass and temperature always have the same average density. b. molecules of different gases with the same mass and temperature always have the same average volume. c. molecules of different gases with the same mass and temperature always have the same pressure. d. molecules of different gases with the same mass and temperature always have the same molecular mass. e. molecules of different gases with the same mass and temperature always have the same average kinetic energy.
Answers: 1
You know the right answer?
Calculate the specific volume of helium using the compressibility factor. 500 kpa, 60 degrees celsiu...
Questions


History, 17.10.2021 14:00

History, 17.10.2021 14:00

French, 17.10.2021 14:00

History, 17.10.2021 14:00

Physics, 17.10.2021 14:00

Physics, 17.10.2021 14:00


Mathematics, 17.10.2021 14:00

Mathematics, 17.10.2021 14:00



Biology, 17.10.2021 14:00

Chemistry, 17.10.2021 14:00

History, 17.10.2021 14:00





Mathematics, 17.10.2021 14:00

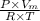
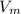 = molar volume
= molar volume