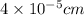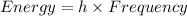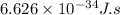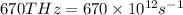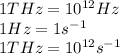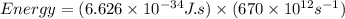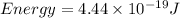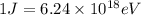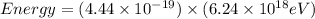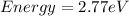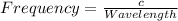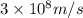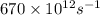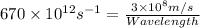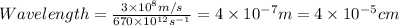Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 22.06.2019 00:30
You have 125g of a certain seasoning and are told that it contains 76.0 g of salt what is the percentage of salt by mass in this seasoning
Answers: 1

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

You know the right answer?
Determine the a) energy (in ev) and b) wavelength (in cm) corresponding to blue light of frequency 6...
Questions

Computers and Technology, 26.11.2020 23:30

Arts, 26.11.2020 23:30

English, 26.11.2020 23:30


History, 26.11.2020 23:30

Computers and Technology, 26.11.2020 23:30

Mathematics, 26.11.2020 23:30

Mathematics, 26.11.2020 23:30

Spanish, 26.11.2020 23:30

History, 26.11.2020 23:30

Physics, 26.11.2020 23:30

Health, 26.11.2020 23:30



English, 26.11.2020 23:30

Mathematics, 26.11.2020 23:30