
Consider the following chemical reaction: co (g) + 2h2(g) ↔ ch3oh(g) at equilibrium in a particular experiment, the concentrations of co and h2 were 0.15 m and0.36 m, respectively. what is the equilibrium concentration of ch3oh? the value of keq for this reaction is 14.5 at the temperature of the experiment.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:10
Agas mixture with a total pressure of 745 mmhg contains each of the following gases at the indicated partial pressures: co2, 245 mmhg ; ar, 119 mmhg ; and o2, 163 mmhg . the mixture also contains helium gas. part a what is the partial pressure of the helium gas? phe p h e = nothing mmhg request answer part b what mass of helium gas is present in a 10.2-l sample of this mixture at 283 k ? m m = nothing g request answer
Answers: 1

Chemistry, 21.06.2019 18:30
150.0 grams of asf3 were reacted with 180.0 g of ccl4 to produce ascl3 and ccl2f2. if the actual yield of ccl2f2 was 127 g, what is the percent yield?
Answers: 2

Chemistry, 23.06.2019 03:00
Analyze the reaction to determine whether the reaction is exothermic or endothermic. explain your reasoning.
Answers: 1

Chemistry, 23.06.2019 05:00
1. true or false: minerals are inorganic. true false 2. inorganic means that something has never been found alive 3. halite is another name for and is a mineral with a cubic crystal pattern. table salt rock salt
Answers: 2
You know the right answer?
Consider the following chemical reaction: co (g) + 2h2(g) ↔ ch3oh(g) at equilibrium in a particular...
Questions

Mathematics, 21.08.2019 19:00


Mathematics, 21.08.2019 19:00

English, 21.08.2019 19:00


Mathematics, 21.08.2019 19:00






History, 21.08.2019 19:00

History, 21.08.2019 19:00

Mathematics, 21.08.2019 19:00




English, 21.08.2019 19:00


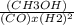 =14.5
=14.5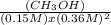
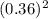 = (CH₃OH)
= (CH₃OH)

