Chemistry, 18.09.2019 05:20 stephaniero6
Asample of liquefied natural, lng, from alaska has the following molar composition: 3.9% c2h6, 1.1% c3h8, 1.1% co2, and the rest ch4. determine (a) the average molecular weight of the lng mixture. (b) the weight fraction of ch4 in the mixture. (c) the lng is heated to 422 k and 148 kpa, and vaporizes completely. estimate the density of the gas mixture under these conditions.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 12:00
the mississippians were considered to be horticulturalists, which means they were
Answers: 1

Chemistry, 22.06.2019 12:30
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2
You know the right answer?
Asample of liquefied natural, lng, from alaska has the following molar composition: 3.9% c2h6, 1.1%...
Questions



History, 23.04.2020 13:46



Mathematics, 23.04.2020 13:48

Mathematics, 23.04.2020 13:48

Biology, 23.04.2020 13:48



Mathematics, 23.04.2020 13:49


Mathematics, 23.04.2020 13:49

German, 23.04.2020 13:50

Mathematics, 23.04.2020 13:50

Mathematics, 23.04.2020 13:50

English, 23.04.2020 13:50


Mathematics, 23.04.2020 13:51

Mathematics, 23.04.2020 13:51

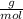
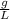
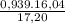 ×100 = 87,57%
×100 = 87,57%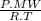 = δ
= δ