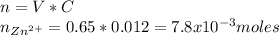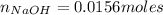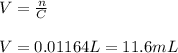Chemistry, 13.09.2019 04:20 ashtor1943
If you had a 0.650 l solution containing 0.0120 m of zn2+(aq), and you wished to add enough 1.34 m naoh(aq) to precipitate all of the metal, what is the minimum amount of the naoh(aq) solution you would need to add? assume that the naoh(aq) solution is the only source of oh−(aq) for the precipitation.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
The pressure in a fluid is affected by which characteristics of that fluid
Answers: 1

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

You know the right answer?
If you had a 0.650 l solution containing 0.0120 m of zn2+(aq), and you wished to add enough 1.34 m n...
Questions




History, 08.09.2020 14:01

Social Studies, 08.09.2020 14:01


Arts, 08.09.2020 14:01

Chemistry, 08.09.2020 14:01


Business, 08.09.2020 14:01


Computers and Technology, 08.09.2020 14:01


Mathematics, 08.09.2020 14:01

Computers and Technology, 08.09.2020 14:01


Computers and Technology, 08.09.2020 14:01