

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:10
Which class of molecules functions as chemical signals? hormones water carbohydrates proteins
Answers: 1


Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3
You know the right answer?
2.4 bromium has two naturally occurring isotopes: 79br, with an atomic weight of 78.918 amu, and 81...
Questions

Mathematics, 20.09.2019 00:10


History, 20.09.2019 00:10

Arts, 20.09.2019 00:10

History, 20.09.2019 00:10





History, 20.09.2019 00:10










Mathematics, 20.09.2019 00:10

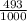 81Br and
81Br and 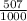 79BR
79BR

