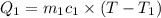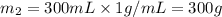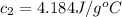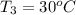Chemistry, 07.09.2019 03:20 miguearrobita
A20g sample of iron at a temperature of 120oc is placed into a container of water. there are 300 milliliters of water in the container at a temperature of 30oc. ciron=0.444jg∘c cwater=4.184jg∘c rhowater=1gml 10. what is the final temperature of the water?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Where are each of the three particles located within the atom?
Answers: 1

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3

Chemistry, 23.06.2019 07:30
Which statement is actually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 2
You know the right answer?
A20g sample of iron at a temperature of 120oc is placed into a container of water. there are 300 mil...
Questions



Mathematics, 18.01.2020 07:31







Chemistry, 18.01.2020 07:31

Mathematics, 18.01.2020 07:31



Mathematics, 18.01.2020 07:31




English, 18.01.2020 07:31


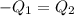
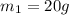
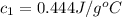
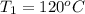
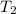 =T
=T