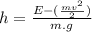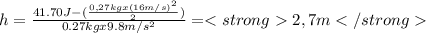Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3


Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1
You know the right answer?
Elias serves a volleyball at a velocity of 16 m/s. the mass of the volleyball is 0.27 kg. what is th...
Questions

Mathematics, 11.02.2021 06:10


History, 11.02.2021 06:10

English, 11.02.2021 06:10


Mathematics, 11.02.2021 06:10

History, 11.02.2021 06:10

Mathematics, 11.02.2021 06:10

Social Studies, 11.02.2021 06:10

Geography, 11.02.2021 06:10

Mathematics, 11.02.2021 06:10

Mathematics, 11.02.2021 06:10

Mathematics, 11.02.2021 06:10


Spanish, 11.02.2021 06:10



English, 11.02.2021 06:10


Health, 11.02.2021 06:10

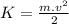 and potential energy like
and potential energy like 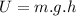 Where m is the mass, v is the velocity, g is the gravity on the earth surface (9,8m/s^2) and h is the height we want to know, so:
Where m is the mass, v is the velocity, g is the gravity on the earth surface (9,8m/s^2) and h is the height we want to know, so: