
Chemistry, 05.09.2019 16:10 dorothybean
The combustion of propane (c3h8) produces co2 and h2o: c3h8(g) 5o2(g)→3co2(g) 4h2o(g) the reaction of 2.5 mol of o2 with 4.6 mol of c3h8 will produce mol of h2o.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 23:30
Why do oxygen have a strong attractive force for electrons
Answers: 2

Chemistry, 23.06.2019 02:50
For questions 1 and 2, consider the following experimental data.hydrogen emission lines were detected at the following wavelengths (in nm): 121.6102.697.395.093.8question 1use the electromagnetic radiation classifications below and figure 1-1 in the introductory information for this lab (in the lab manual) to determine the nf value for the experimental data provided? wavelength, ? (nm) 650 700 550 600 400 450 500 visible spectrum wavelength, ? (m) 11 10 3 10 10 10 8 10 5 10 10 -10 10 9 10 10 10 10 -12 10 microwave radio infrared x-ray ultraviolet gamma 1020 1019 1018 1 1016 015 1014 01 12 109108 frequency, v (hz)a.1b. 2c. 3d. 4e. 5question 2using the data for the emission line with the longest wavelength, the known value of nf (from question 1 in this prelab), and the value of ni (deduced from the ? and nf values) calculate the rydberg constant for hydrogen (rh) in units of m-1.a) 1.097 x 10-11 m-1b) 5.921 x 107 m-1c) 1.097 x 10-2 m-1d) 9.252 x 106 m-1e) 1.097 x 107 m-1
Answers: 3
You know the right answer?
The combustion of propane (c3h8) produces co2 and h2o: c3h8(g) 5o2(g)→3co2(g) 4h2o(g) the reaction...
Questions

English, 30.06.2021 22:50

Mathematics, 30.06.2021 22:50







History, 30.06.2021 22:50

Mathematics, 30.06.2021 22:50

Mathematics, 30.06.2021 22:50

Mathematics, 30.06.2021 22:50

History, 30.06.2021 22:50


Mathematics, 30.06.2021 22:50

Mathematics, 30.06.2021 22:50

Social Studies, 30.06.2021 22:50


Mathematics, 30.06.2021 22:50

History, 30.06.2021 22:50

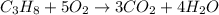
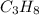 = 4.6
= 4.6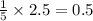 moles of oxygen
moles of oxygen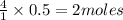 of water
of water

