
Chemistry, 03.09.2019 16:30 jholland03
Joseph priestley is credited with the discovery of oxygen. he pro- duced o2 by heating mercury(ii) oxide, hgo, to decompose it into its elements. how many moles of oxygen could priestley have pro- duced if he had decomposed 517.84 g of mercury oxide?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Consider the balanced equation below. n2h4 + 2h2o2 n2 + 4h2o what are the mole ratios of hydrazine (n2h4) to hydrogen peroxide (h2o2) and hydrazine to water? 1: 2 and 1: 4 1: 3 and 1: 4 1: 2 and 3: 5 1: 3 and 3: 5
Answers: 3

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2


Chemistry, 22.06.2019 15:30
The identities of substances are the same before and after which type of change
Answers: 1
You know the right answer?
Joseph priestley is credited with the discovery of oxygen. he pro- duced o2 by heating mercury(ii) o...
Questions

Biology, 18.03.2020 05:04

Mathematics, 18.03.2020 05:04







Mathematics, 18.03.2020 05:05

Mathematics, 18.03.2020 05:05




History, 18.03.2020 05:05






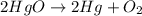
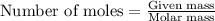
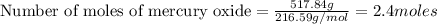
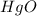 gives 1 mole of
gives 1 mole of 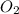
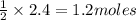 of
of 


