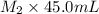Chemistry, 02.09.2019 19:10 suzymott1562
What is the final concentration of the solution produced when 225.5 ml of 0.09988-m solution of na2co3 is allowed to evaporate until the solution volume is reduced to 45.00 ml?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 21.06.2019 23:10
Nitrogen (n), phosphorus (p), and potassium (k) are the main nutrients in plant fertilizers. according to an industry convention, the numbers on the label refer to the mass percents of n, p2o5, and k2o, in that order. calculate the n: p: k ratio of a 30: 10: 10 fertilizer in terms of moles of each element, and express it as x: y: 1.0.
Answers: 1

Chemistry, 22.06.2019 07:00
6what is the importance of water on earth? a) it keeps the top layer of the geosphere cool b) it allows life to exist c) it provides ice at the poles d) it creates earth's blue color from space
Answers: 2

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1
You know the right answer?
What is the final concentration of the solution produced when 225.5 ml of 0.09988-m solution of na2c...
Questions

Mathematics, 03.09.2020 19:01


Mathematics, 03.09.2020 19:01

Biology, 03.09.2020 19:01



Mathematics, 03.09.2020 19:01

Mathematics, 03.09.2020 19:01



Biology, 03.09.2020 19:01

Mathematics, 03.09.2020 19:01

Computers and Technology, 03.09.2020 19:01

Social Studies, 03.09.2020 19:01



Mathematics, 03.09.2020 19:01


Mathematics, 03.09.2020 19:01

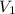 = 225.5 mL,
= 225.5 mL, 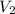 = 45.00 mL
= 45.00 mL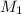 = 0.09988 M,
= 0.09988 M, 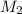 = ?
= ?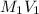 =
= 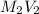
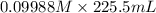 =
=