Chemistry, 30.08.2019 18:10 MissSmartyPants88
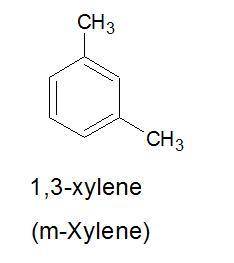
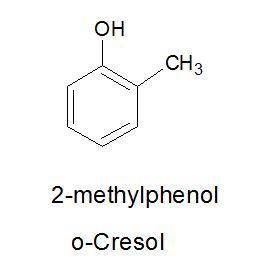
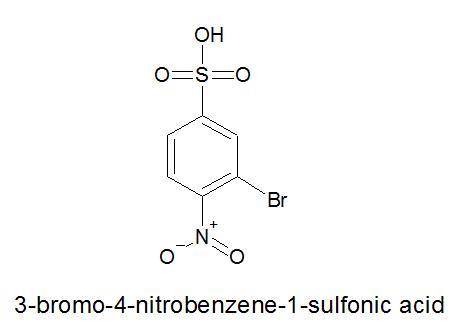
Draw structure for each of the following a) m-xylene b) o-cresol c) 3-bromo-4-nitrobenzenesulfonic acid

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
Draw structure for each of the following a) m-xylene b) o-cresol c) 3-bromo-4-nitrobenzenesulfonic a...
Questions

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

English, 16.09.2020 01:01