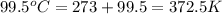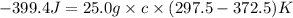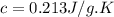A25.0g tin sample was placed in boiling water at 99.5 c until it had the same temperature. afterwards, the tin sample was placed in room temperature water. if the tin sample lost 399.4j of heat and its final temperature was 24.5 c, what is the specific heat of tin?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Which is a chemical property of iron? a. it forms iron oxide (rust) when exposed to moisture and air. b. it is a gray–black metal that is hard to the touch. c. it has a melting point of 2795°f (1536°c). d. it is a good conductor of heat
Answers: 2

Chemistry, 22.06.2019 08:30
Since the gas in your graduated cylinder is a mixture of butane and water vapor, you must determine the partial pressure of the butane, pbutane, alone. to do this, consult a reference and record the partial pressure of the water vapor, pwater, at the temperature you recorded. use the following formula to compute the partial pressure of the butane. pbutane = atmosphere - pwater use the following combined gas law formula and compute the volume that the butane sample will occupy at stp. (hint: convert both temperatures to kelvin.) pbutane x voriginal = pstandard x vfinal troom tstandard use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. grams of butane you used “x” grams of butane ml of butane corrected to stp = 22,400 ml compute the theoretical molar mass of butane based on its formula and the atomic masses on the periodic table. compare your experimental results from #3 to the theoretical value of #4, computing a percent error of your findings using this formula: % error = measured value - accepted value x 100 accepted value use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. need asap
Answers: 1


Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1
You know the right answer?
A25.0g tin sample was placed in boiling water at 99.5 c until it had the same temperature. afterward...
Questions

Mathematics, 04.03.2021 18:10



English, 04.03.2021 18:10


Mathematics, 04.03.2021 18:10



History, 04.03.2021 18:10

English, 04.03.2021 18:10


Mathematics, 04.03.2021 18:10





History, 04.03.2021 18:10

Advanced Placement (AP), 04.03.2021 18:10

Mathematics, 04.03.2021 18:10

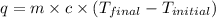
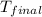 = final temperature =
= final temperature = 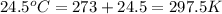
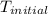 = initial temperature =
= initial temperature =