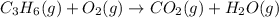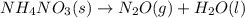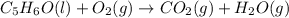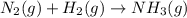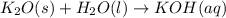Chemistry, 28.08.2019 17:20 mauricio18s
Balance the following equations, and indicate whether they are combination, decomposition, or combustion reactions.1. c3h6(g)+o2(> co2(g)+h2o(g)1a. is it a combination reaction, decomposition reaction, or combustion reaction2. nh4no3(> n2o(g)+h2o(l)2a. is it a combination reaction, decomposition reaction, or combustion reaction3. c5h6o(l)+o2(> co2(g)+h2o(g)3a. is it a combination reaction, decomposition reaction, or combustion reaction4.n2(g)+h2(> nh3(g)? 4a. is it a combination reaction, decomposition reaction, or combustion reaction5. k2o(s)+h2o(> koh(aq)5a. is it a combination reaction, decomposition reaction, or combustion reaction

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:10
Here’s one way to follow the scientific method. place the missing steps in the correct position in the process
Answers: 1

Chemistry, 22.06.2019 19:00
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
You know the right answer?
Balance the following equations, and indicate whether they are combination, decomposition, or combus...
Questions



English, 05.05.2021 16:20

Mathematics, 05.05.2021 16:20


Mathematics, 05.05.2021 16:20




Mathematics, 05.05.2021 16:20

Mathematics, 05.05.2021 16:20




Mathematics, 05.05.2021 16:20


English, 05.05.2021 16:20


Mathematics, 05.05.2021 16:20