
Chemistry, 27.08.2019 16:30 brandy2115
Na2co3 + hcl → co2 + h2o + naclconsider the above unbalanced equation. for this reaction, how many ml of a 2 m solution of na2co3 are required to produce 11.2 l of co2 at stp?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 23.06.2019 08:00
How does the digestive system interact with the circulatory system? a. messages sent as electrical impulses from the digestive system are transported throughout the body by the circulatory system. b. nutrients taken in and broken down by the digestive system are carried to various parts of the body by the circulatory system. c. nutrients and gases are absorbed by organs in the circulatory system. then, they are transported to all parts of the body by organs in the digestive system. d. oxygen and carbon dioxide are exchanged by organs in the digestive system, and the gases are carried to the rest of the body by the circulatory system.
Answers: 2

Chemistry, 23.06.2019 10:30
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 2
You know the right answer?
Na2co3 + hcl → co2 + h2o + naclconsider the above unbalanced equation. for this reaction, how many m...
Questions

Mathematics, 06.10.2019 21:00

Social Studies, 06.10.2019 21:00

Mathematics, 06.10.2019 21:00


Spanish, 06.10.2019 21:00


Business, 06.10.2019 21:00

Business, 06.10.2019 21:00

Mathematics, 06.10.2019 21:00


Mathematics, 06.10.2019 21:00


History, 06.10.2019 21:00


Mathematics, 06.10.2019 21:00


Mathematics, 06.10.2019 21:00

English, 06.10.2019 21:00

English, 06.10.2019 21:00

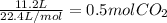
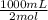 = 250 mL
= 250 mL

