
Chemistry, 23.08.2019 04:30 elizabethburkha
An electrochemical cell at 25°c is composed of pure copper and pure lead solutions immersed in their respective ionis. for a 0.6 m concentration of cu2+, the lead electrode is oxidized yielding potential of 0.507 v. a cell a) calculate the concentration of pb2+ b) suppose the lead electrode is reduced, in that case what would be the concentration of pb2 what does this answer tell you?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 13:30
What produces wave a)sound b) heats c)transfer of energy d)vibrations
Answers: 2

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 15:30
The identities of substances are the same before and after which type of change
Answers: 1
You know the right answer?
An electrochemical cell at 25°c is composed of pure copper and pure lead solutions immersed in their...
Questions

Biology, 19.10.2019 04:00




Social Studies, 19.10.2019 04:00



English, 19.10.2019 04:00

Physics, 19.10.2019 04:00

History, 19.10.2019 04:00

Computers and Technology, 19.10.2019 04:00

English, 19.10.2019 04:00

Mathematics, 19.10.2019 04:00


Biology, 19.10.2019 04:00


Mathematics, 19.10.2019 04:00

Mathematics, 19.10.2019 04:00

English, 19.10.2019 04:00

Biology, 19.10.2019 04:00

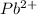 is, 0.0337 M
is, 0.0337 M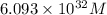
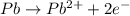
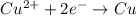
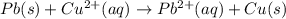
![E^o_{[Pb^{2+}/Pb]}=-0.13V](/tpl/images/0189/9678/d356c.png)
![E^o_{[Cu^{2+}/Cu]}=+0.34V](/tpl/images/0189/9678/0d165.png)
![E^o=E^o_{[Cu^{2+}/Cu]}-E^o_{[Pb^{2+}/Pb]}](/tpl/images/0189/9678/2597c.png)
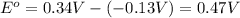
![E_{cell}=E^o_{cell}-\frac{0.0592}{n}\log \frac{[Pb^{2+}]}{[Cu^{2+}]}](/tpl/images/0189/9678/2e714.png)
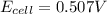
![0.507=0.47-\frac{0.0592}{2}\log \frac{[Pb^{2+}]}{(0.6)}](/tpl/images/0189/9678/33110.png)
![[Pb^{2+}]=0.0337M](/tpl/images/0189/9678/42065.png)
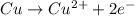
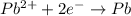
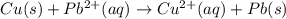
![E^o=E^o_{[Pb^{2+}/Pb]}-E^o_{[Cu^{2+}/Cu]}](/tpl/images/0189/9678/264d0.png)
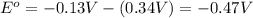
![E_{cell}=E^o_{cell}-\frac{0.0592}{n}\log \frac{[Cu^{2+}]}{[Pb^{2+}]}](/tpl/images/0189/9678/9bec7.png)
![0.507=-0.47-\frac{0.0592}{2}\log \frac{(0.6)}{[Pb^{2+}]}](/tpl/images/0189/9678/15aa8.png)
![[Pb^{2+}]=6.093\times 10^{32}M](/tpl/images/0189/9678/e8ea8.png)


