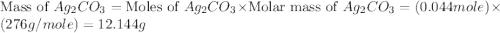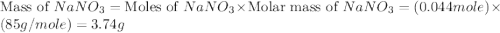Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 4.25 g of sodium carbonate is mixed with one containing 7.50 g of silver nitrate. how many grams of each of the following compounds are present after the reaction is complete?
i) sodium carbonate
ii) silver nitrate
iii) silver carbonate
iv) sodium nitrate

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1

Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 2
You know the right answer?
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution...
Questions

Geography, 02.07.2019 11:00

Chemistry, 02.07.2019 11:00

English, 02.07.2019 11:00

English, 02.07.2019 11:00

Mathematics, 02.07.2019 11:00


Mathematics, 02.07.2019 11:00


English, 02.07.2019 11:00

English, 02.07.2019 11:00


History, 02.07.2019 11:00


Physics, 02.07.2019 11:00

Biology, 02.07.2019 11:00



Physics, 02.07.2019 11:00


Chemistry, 02.07.2019 11:00

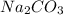 ,
, 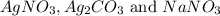 are, 1.908 g, 0 g, 12.144 g and 3.74 g respectively.
are, 1.908 g, 0 g, 12.144 g and 3.74 g respectively.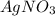 = 7.50 g
= 7.50 g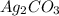 = 276 g/mole
= 276 g/mole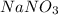 = 85 g/mole
= 85 g/mole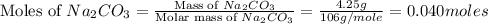
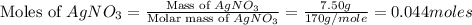
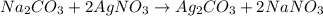
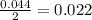 moles of
moles of 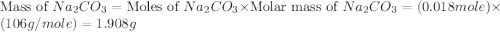
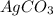 .
.