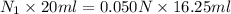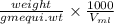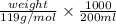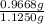Chemistry, 22.08.2019 00:20 chrisdelreal86
Asample containing kbr (m 119& /mol) and kno3 (m 101 g/mol) was analysed by using the mohr method, a 1.1250-g sample is dissolved into 200.0 ml solution (solution a). 20.00-ml. of solution a was titrated with 16.25 ml of 0.oson agnos (m 170) solution. a) report c of ca solution & according to the tination data b) calculate c% w/w of kbr in the original sample.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2


Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2
You know the right answer?
Asample containing kbr (m 119& /mol) and kno3 (m 101 g/mol) was analysed by using the mohr metho...
Questions


Mathematics, 19.03.2021 21:20


History, 19.03.2021 21:20


Social Studies, 19.03.2021 21:20



Mathematics, 19.03.2021 21:20

History, 19.03.2021 21:20

Mathematics, 19.03.2021 21:20

English, 19.03.2021 21:20

Mathematics, 19.03.2021 21:20

Mathematics, 19.03.2021 21:20


Mathematics, 19.03.2021 21:20




Mathematics, 19.03.2021 21:20

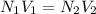
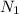 = ?,
= ?, 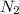 = 0.050 N
= 0.050 N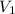 = 20 ml,
= 20 ml, 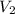 = 16.25 ml
= 16.25 ml