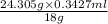The solubility of water in diethyl ether has been reported to be 1.468 % by mass.' assuming that 23.0 ml of diethyl ether were allowed to become saturated with water before used and that the 1.2 g magnesium was the limiting reagent, what percentage of the product is expected to be lost due to the water in the solvent if the ether is not anhydrous?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 22.06.2019 22:30
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3

Chemistry, 23.06.2019 01:00
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1
You know the right answer?
The solubility of water in diethyl ether has been reported to be 1.468 % by mass.' assuming that 23....
Questions

History, 28.01.2020 16:41




Biology, 28.01.2020 16:41


Social Studies, 28.01.2020 16:41



Mathematics, 28.01.2020 16:42





Mathematics, 28.01.2020 16:42


History, 28.01.2020 16:42



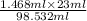
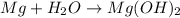
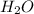 = 18 g
= 18 g