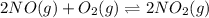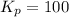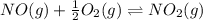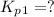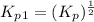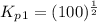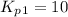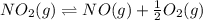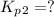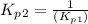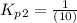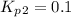Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
After cloud droplets form, what must happen to them for precipitation to occur?
Answers: 1

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 14:30
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1

You know the right answer?
Find the equilibrium constants, kp, for the following equilibria, (i) no(g) + ½ o2(g) ⇄ no2(g), kp =...
Questions


Biology, 16.10.2020 09:01

History, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01




Mathematics, 16.10.2020 09:01

Social Studies, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01


English, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01

Arts, 16.10.2020 09:01



History, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01