

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
One mole of zinc has a mass of 65.4 grams. approximately how many atoms of zinc are present in one mole of zinc? 32 × 1023 atoms 6 × 1023 atoms 66 atoms 65 atoms
Answers: 1

Chemistry, 22.06.2019 00:40
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2

Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2
You know the right answer?
Which of the following is not an acid-base conjugate pair?
a. nh4+ and nh3
b. h2o and...
a. nh4+ and nh3
b. h2o and...
Questions


Computers and Technology, 15.01.2021 20:00

Mathematics, 15.01.2021 20:00

Biology, 15.01.2021 20:00

Chemistry, 15.01.2021 20:00





Social Studies, 15.01.2021 20:00



Social Studies, 15.01.2021 20:00

Mathematics, 15.01.2021 20:00




World Languages, 15.01.2021 20:00

Chemistry, 15.01.2021 20:00

Mathematics, 15.01.2021 20:00

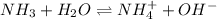
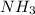 and
and 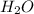 are acid and base and
are acid and base and 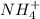 and
and 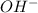 are conjugate acid and base respectively.
are conjugate acid and base respectively.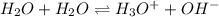
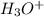 and
and 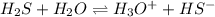
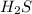 and
and 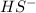 are conjugate acid and base respectively.
are conjugate acid and base respectively.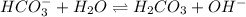
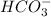 and
and 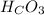 and
and 

