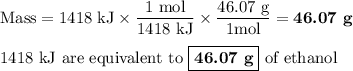Chemistry, 18.08.2019 14:10 rleiphart1
The equation shows one mole of ethanol fuel being burned in oxygen. convert the energy released into its equivalent mass. c2h5oh(l) + 3 o2(g) → 2 co2(g) + 3 h2o (l) δh = -1418 kj/mol

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1
You know the right answer?
The equation shows one mole of ethanol fuel being burned in oxygen. convert the energy released into...
Questions

Mathematics, 10.02.2021 01:50



English, 10.02.2021 01:50

Mathematics, 10.02.2021 01:50

History, 10.02.2021 01:50

Mathematics, 10.02.2021 01:50



Mathematics, 10.02.2021 01:50

Health, 10.02.2021 01:50

Mathematics, 10.02.2021 01:50


Computers and Technology, 10.02.2021 01:50


Mathematics, 10.02.2021 01:50

Social Studies, 10.02.2021 01:50

Mathematics, 10.02.2021 01:50


English, 10.02.2021 01:50